Base-Catalyzed Aryl-B(OH)2 Protodeboronation Revisited: From

Base-Catalyzed Aryl-B(OH)2 Protodeboronation Revisited: From

Protodeboronation of 5-formyl-2-thiopheneboronic acid

Base-Catalyzed Aryl-B(OH)2 Protodeboronation Revisited: From Concerted Proton Transfer to Liberation of a

Base-Catalyzed Aryl-B(OH)2 Protodeboronation Revisited: From Concerted Proton Transfer to Liberation of a Transient Aryl Anion

Figure 11 from Base-Catalyzed Aryl-B(OH)2 Protodeboronation Revisited: From Concerted Proton Transfer to Liberation of a Transient Aryl Anion.

Mechanism of Cu-Catalyzed Aryl Boronic Acid Halodeboronation Using Electrophilic Halogen: Development of a Base-Catalyzed Iododeboronation for Radiolabeling Applications

Base-free nickel-catalysed decarbonylative Suzuki-Miyaura coupling of acid fluorides. - Abstract - Europe PMC

Development and Molecular Understanding of a Pd‐Catalyzed Cyanation of Aryl Boronic Acids Enabled by High‐Throughput Experimentation and Data Analysis - De Jesus Silva - 2021 - Helvetica Chimica Acta - Wiley Online Library
The proposed mechanism for protodeboronation of arylboronic acids

Activator-regulated chemodivergent deoxygenative- and alkoxy-carbonylation of alcohols with boronic acids - ScienceDirect

Strategies for successful Suzuki-Miyaura cross-couplings with thienylboronic acids: From model studies to dye structures - ScienceDirect

Non-innocent electrophiles go beyond Research Communities by Springer Nature

Base-Catalyzed Aryl-B(OH)2 Protodeboronation Revisited: From Concerted Proton Transfer to Liberation of a Transient Aryl Anion
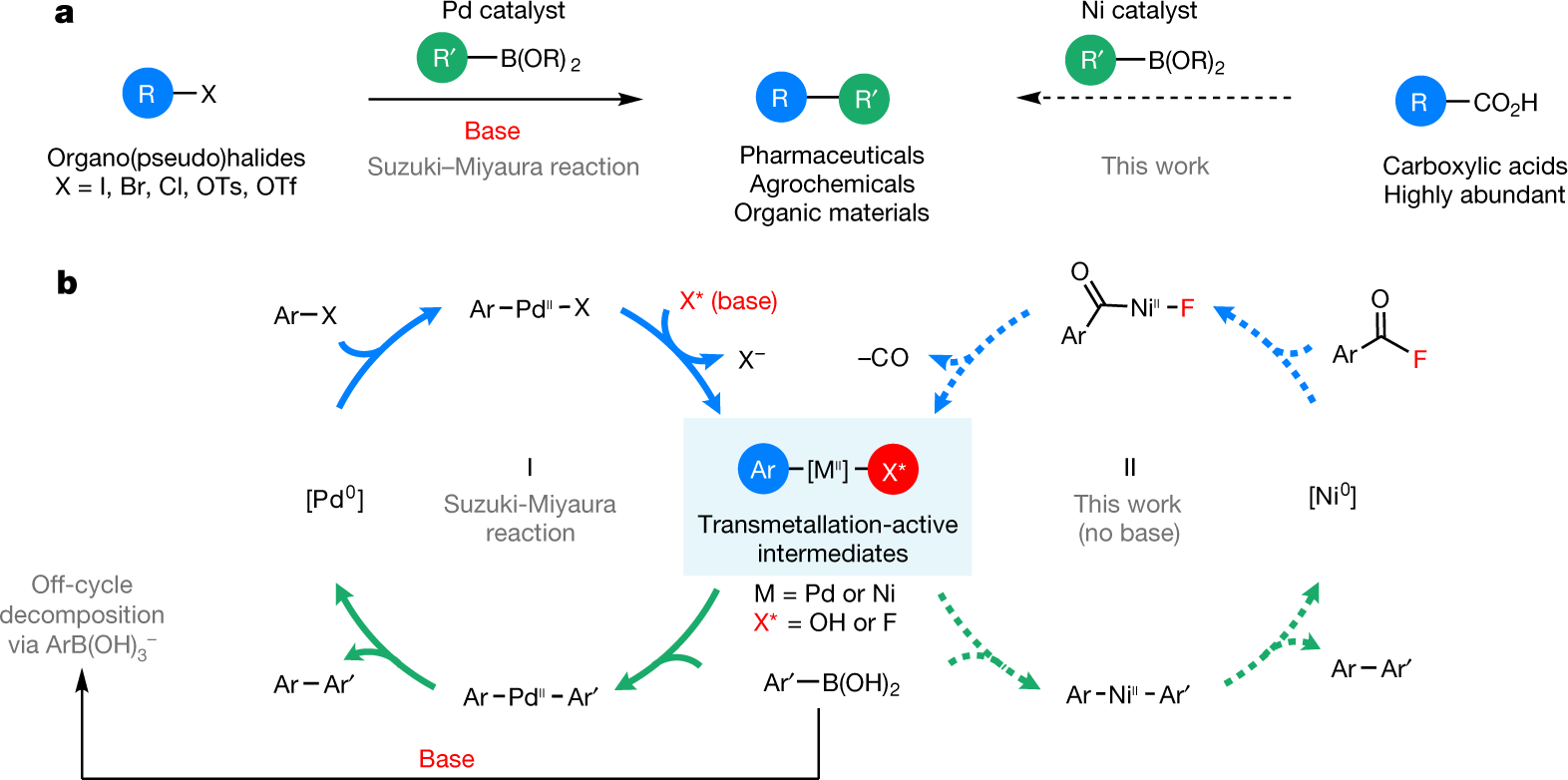
Base-free nickel-catalysed decarbonylative Suzuki–Miyaura coupling of acid fluorides

PDF] Protodeboronation of Heteroaromatic, Vinyl, and Cyclopropyl Boronic Acids: pH-Rate Profiles, Autocatalysis, and Disproportionation.


:max_bytes(150000):strip_icc()/Jacky-Oh-01-060123-fb612ae420964eaaba62b61775873e3a.jpg)